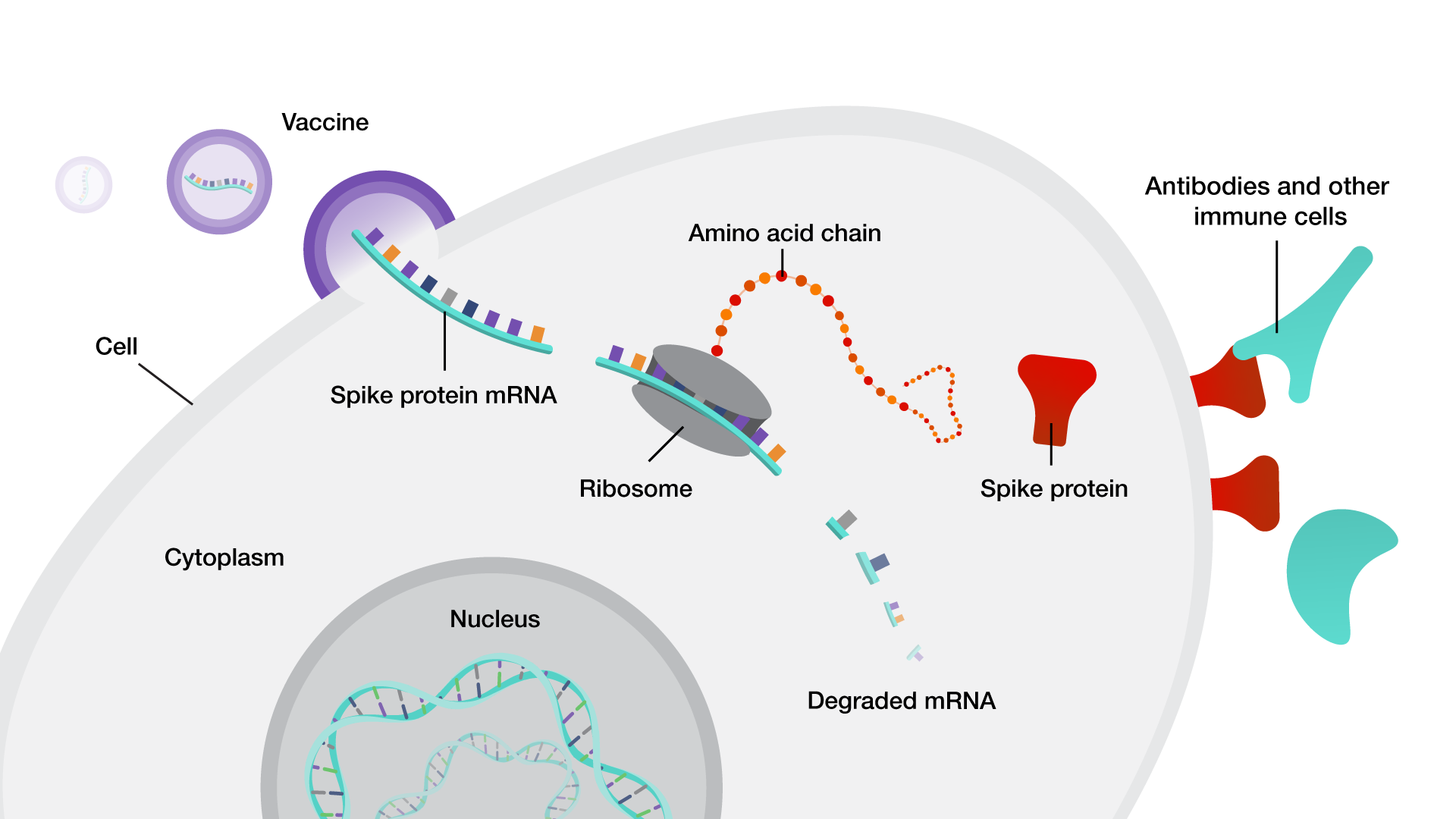 
Ī drawback is that the specific antigens used in a subunit vaccine may lack pathogen-associated molecular patterns which are common to a class of pathogen. Īn effective vaccine would elicit the immune response to the antigens and form immunological memory that allows quick recognition of the pathogens and quick response to future infections. Subunit vaccines contain fragments of the pathogen, such as protein or polysaccharide, whose combinations are carefully selected to induce a strong and effective immune response.īecause the immune system interacts with the pathogen in a limited way, the risk of side effects is minimal. The first certified subunit vaccine by clinical trials on humans is the hepatitis B vaccine, containing the surface antigens of the hepatitis B virus itself from infected patients and adjusted by newly developed technology aiming to enhance the vaccine safety and eliminate possible contamination through individuals plasma. Advice from medical professionals should be sought before receiving any vaccination. The contraindications are also vaccine-specific they are generally not recommended for people with the previous history of anaphylaxis to any component of the vaccines. Minor side effects include injection site pain, fever, and fatigue, and serious adverse effects consist of anaphylaxis and potentially fatal allergic reaction. 
The chances of adverse effects vary depending on the specific type of vaccine being administered. Recombinant subunit vaccines are considered to be safe for injection. Additionally, recombinant subunit vaccines are popular candidates for the development of vaccines against infectious diseases (e.g. Basic components of recombinant subunit vaccines include recombinant subunits, adjuvants and carriers. Notable recombinant subunit vaccines licensed include ENGERIX-B ( hepatitis B), GARDASIL-9 ( Human Papillomavirus), FLUBLOK ( influenza), SHINGRIX ( Herpes Zoster) and NUVAXOVID ( Coronavirus Disease 2019).Īfter injection, antigens trigger the production of antigen-specific antibodies, which are responsible for recognising and neutralising foreign substances. The first recombinant subunit vaccine was produced in the mid-1980s to protect people from Hepatitis B. ĭisadvantages include being relatively complex to manufacture compared to some vaccines, possibly requiring adjuvants and booster shots, and requiring time to examine which antigenic combinations may work best. Other advantages include being well-established technology and being suitable for immunocompromised individuals. īecause the vaccine doesn't contain "live" components of the pathogen, there is no risk of introducing the disease, and is safer and more stable than vaccines containing whole pathogens. 
Subunit vaccine can be made from dissembled viral particles in cell culture or recombinant DNA expression, in which case it is a recombinant subunit vaccine.Ī "subunit" vaccine doesn't contain the whole pathogen, unlike live attenuated or inactivated vaccine, but contains only the antigenic parts such as proteins, polysaccharides or peptides. Vaccine that contains antigenic parts of the pathogen.Ī subunit vaccine is a vaccine that contains purified parts of the pathogen that are antigenic, or necessary to elicit a protective immune response.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
