Therefore the prefix “prop” is used for this. Worked example: Give the name for the following molecule:Įxplanation: The longest carbon chain has three carbon atoms. When there are identical side-chains di- is used for 2, tri- for 3, and tetra- for 4. These are written in alphabetical order and also require the number of the carbon they are attached to. Other functional groups and carbon side- chains are included in the name as prefixes. Include this number as part of the name in front of the suffix e.g. Naming and Representing Organic CompoundsĤ. Ensure that the carbon bonded to the functional group is given a number as small as possible. 
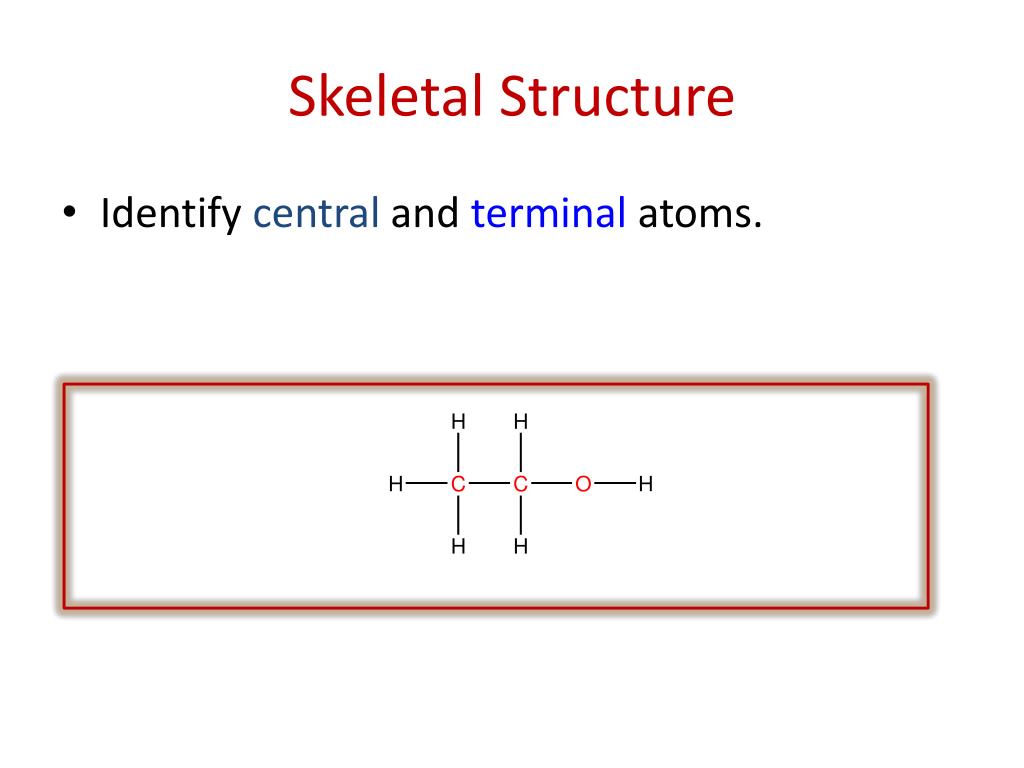
Give the longest chain of carbon numbers. Naming and Representing Organic Compoundsģ. The key functional groups you need to know are: These give us the prefix and suffix of the compound. If there is only one functional group, it will be named as the suffix of the compound. These are the reactive parts of the molecule. Naming and Representing Organic CompoundsĢ. This is key as it will give you the stem of the compound’s name. Determine the length of the longest carbon chain. To name any compound there are some key rules to remember.ġ. Step 2: Multiply the empirical formula by the answer to give the molecular formula.Ĭ) The structural formula is CH3CH2CH2CH2CH2CH2CH2CH2CH2CH3 Mass of empirical formula = (5 x 12)+ (11 x 1) = 71 Step 1: Divide the molecular mass by the mass of the empirical formula. Step 2: Divide by the smallest number of moles to find the simplest ratio Worked example: A hydrocarbon contains 240 g of carbon and 44 g of hydrogen.ī) If the molecular mass is 142, determine its molecular formula.Ī) Step 1: Find the number of moles of each element Naming and Representing Organic Compounds The angle of the C-C bonds in an unbranched chain should be about 109.5°. It shows only a ‘skeleton’ with just functional groups drawn out. It is assumed there are carbon atoms where the bonds meet. The skeletal formula is a representation of organic compound in which the carbon chain is shown by lines with no carbons or hydrogens drawn. For Hexane this would be: CH3CH2CH2CH2CH2CH3 The structural formula is the molecule written with each carbon listed separate. The displayed formula is the fully drawn out version of a molecule. For example the molecular formula of glucose is C6H12O6. The molecular formula is the actual number of atoms of each element present within the molecule. It actually contains 6 x C atoms, 12 x H atoms and 6 X O atoms. For example, the empirical formula of glucose is CH2O. The empirical formula is the simplest ratio of elements in a molecule. Organic compounds are compounds that make up the cells of living organisms and consist mainly of carbon atoms bonded to each other and to atoms of other elements by covalent bonds.Ī functional group is the reactive part of an organic molecule. Introduction to Organic Chemistry - Naming and Representing Organic Compounds (A-Level Chemistry) Organic Compound Nomenclature Key Terms
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
